 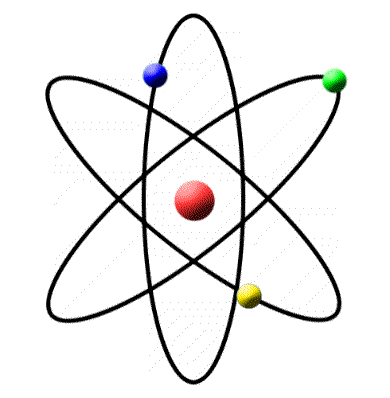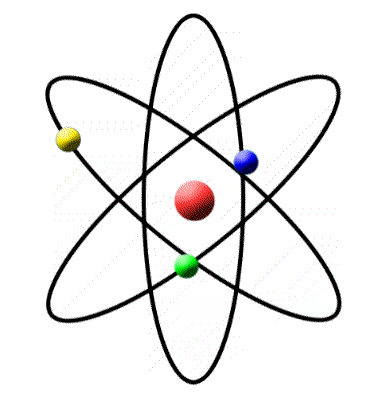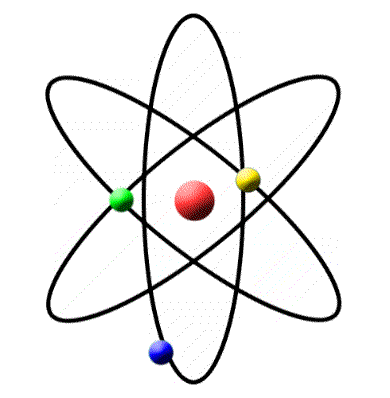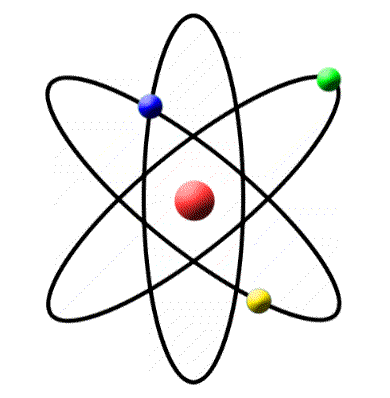
The gaining or losing of electrons, or the sharing of electrons, contributes to the chemical reactions in which an atom participates. Atoms tend to gain or lose electrons until their outer shells have a stable arrangement. The second and all other outer shells have a maximum of eight electrons. The first electron shell has a maximum of two electrons. Gold atoms cannot be broken down into anything smaller while still retaining the properties. For example, a gold coin is simply a very large number of gold atoms molded into the shape of a coin, with small amounts of other, contaminating elements. Atoms are most stable when their outer shell of electrons has a full quota. An atom is the smallest unit of matter that retains all of the chemical properties of an element. The electron arrangement in an atom plays an essential role in the chemistry of the atom. Protons and neutrons adhere tightly to form the dense, positively charged nucleus of the atom. A third type of particle, a neutron, has no electrical charge it has the same weight as a proton. 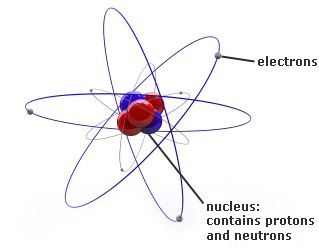
An atom is the smallest part of an element that can enter into combinations with atoms of other elements.Ītoms consist of positively charged particles called protons surrounded by negatively charged particles called electrons. Oxygen, iron, calcium, sodium, hydrogen, carbon, and nitrogen are examples of elements.Įach element is composed of one particular kind of atom. An element is a substance that cannot be chemically decomposed. More than 100 elements are known to exist, including those that are man-made. They soon realized that the chemical organization of all living things is remarkably similar.Īll living things on Earth are composed of fundamental building blocks of matter called elements. Then in the 1700s, scientists discovered the chemical and physical bases of living things. Biologists searched for unidentified plants and animals, classified them, and studied their anatomy and how they acted in nature. Introduction to Nutrition and Digestionįor many centuries, biology was the study of the natural world.Not all the atoms of an element need have the same number of neutrons in their nuclei. Vascular Plants: Structure and Function The periodic table of the elements assigns one place to every atomic number, and each of these places is labeled with the common name of the element, as, for example, calcium, radon, or uranium.The atomic number is 6 since there are 6 protons. Luckily, one electron has the same charge (with opposite sign) as a proton. Quiz Domains and Kingdoms of Living Things A neutral atom is an atom where the charges of the electrons and the protons balance.Quiz Structure of Prokaryote and Eukaryote Cells.The Structure of Prokaryote and Eukaryote Cells.Quiz Movement through the Plasma Membrane. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
